Stefan Constantinescu
Institut de Duve Avenue Hippocrate 75 - B1.74.03 1200 Bruxelles
The Constantinescu Lab studies how blood is formed under the influence of small proteins called cytokines and how blood cancers can be targeted.
Major achievements
- Identification and characterization of JAK2/MPL driver mutations in myeloproliferative neoplasms, and first report of oncogenic JAK1 and TYK2 mutants.
- Identification of orientation-dependent signaling by dimeric cytokine receptors dependent on membrane domains and short cytosolic helical motifs.
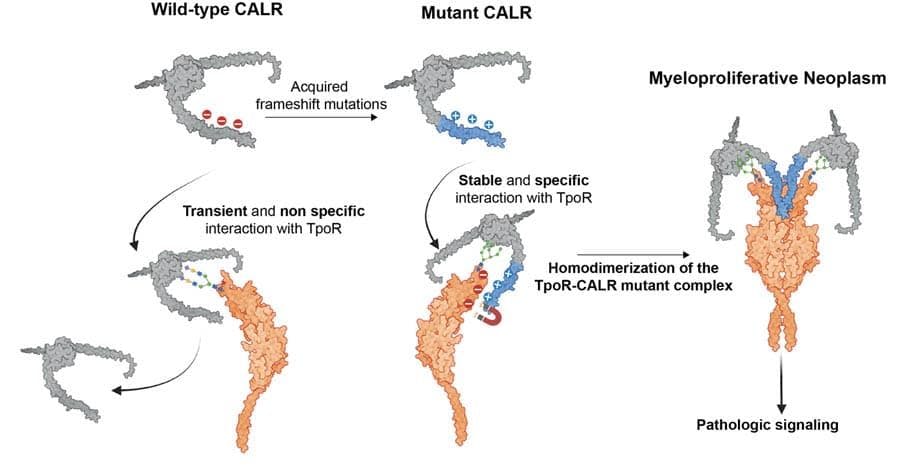
- Discovery of the mechanism by which mutated calreticulins induce blood cancer as first example of a rogue chaperones becoming an oncoprotein.
Research
Question
How do activated mutants of cytokine receptors and JAK-STAT proteins trigger blood cancers, and how do they interact with epigenetic regulators to promote clonal expansion?
Research focus
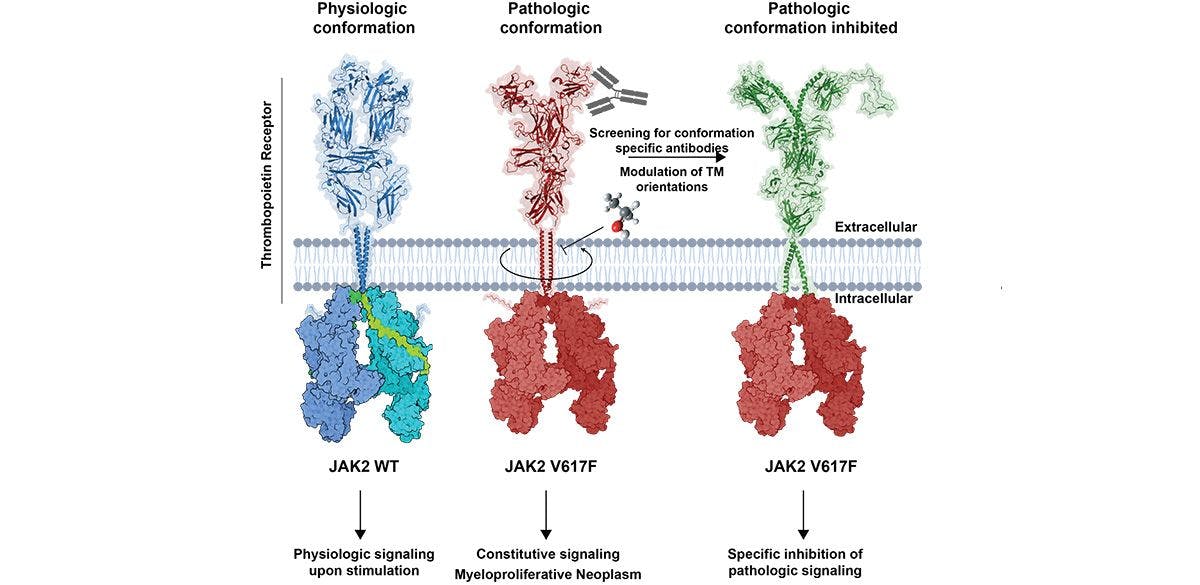
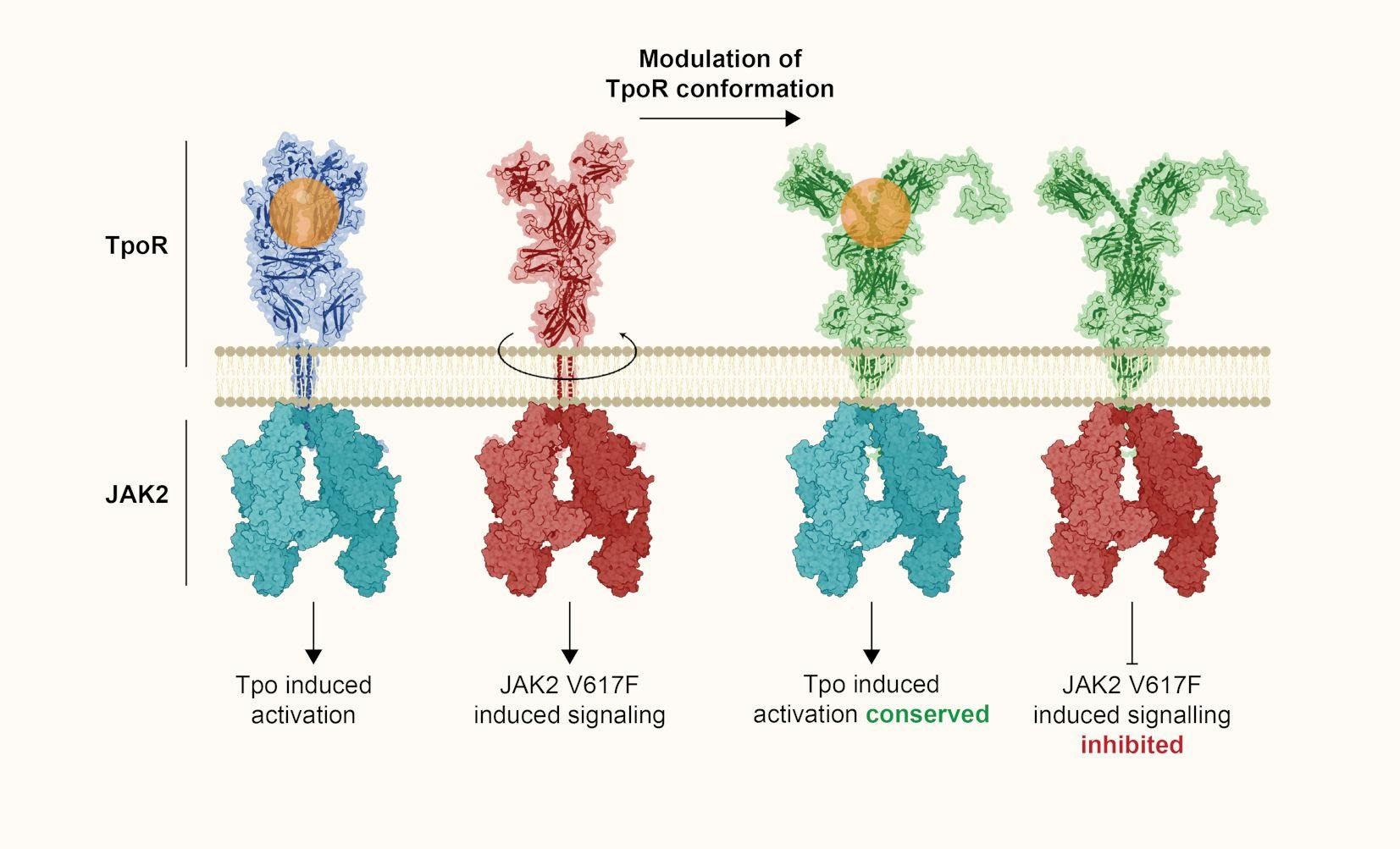
The identification that the thrombopoietin receptor adopts different conformations when it is attached to wild-type JAK2 (left) or to the mutant JAK2 V617F (middle). This may allow the development of novel specific therapies (right) that would prevent the pathologic conformation induced by JAK2 V617F.
Formation of blood requires small circulating proteins, called cytokines, such as erythropoietin, or thrombopoietin that induce survival, growth and differentiation of blood precursors. They bind to receptors on target cells, which function like ‘antennae’ that transmit a signal to the cell interior. We study how these specific receptors assemble on the cell membrane and couple at the cell interior to JAKs, which are absolutely required to transmit a signal. We found that mutations in JAKs or in receptors themselves or in proteins that can activate receptors (such as the chaperone calreticulin) confuse the cells and make them grow indefinitely, leading to blood cancers, specifically myeloproliferative neoplasms (MPNs). A curative treatment needs mutant-specific inhibitors. We found that the conformation of receptors or JAK2 mutants differ from physiologic receptor-JAK2 complexes, which may allow targeted inhibition (Figure). We study how signaling mutations impact epigenetic regulators and how MPNs progress to acute myeloid leukemia. We also study leukemia in children, especially those being resistant to treatment. This work is supported by ‘Les avions de Sébastien’.
In order to pursue our aims, we use molecular, biophysical approaches, like deuterium exchange mass spectrometry as well as in vivo transgenesis, mouse bone marrow transplantation, as well as investigation of primary patient cells.
Projects
- IFNα targeting JAK2 V617F-positive myeloid cancers
- Targeting mechanisms of pathogenic dimerization and activation of thrombopoietin receptor by mutant calreticulins and by JAK2 V617F in myeloproliferative neoplasms
Diseases
Bio
Stefan Constantinescu obtained his MD (1988) and PhD in Virology (1991) at Carol Davila University of Medicine and Pharmacy Bucharest. As a PhD student while in training in adult and pediatric hemato-oncology he discovered an epidemy of pediatric AIDS in children in Romania (Lancet 1990, 335, 672), which had public health impact. He performed postdoctoral studies at Whitehead Institute for Biomedical Research at MIT, Cambridge MA, USA (with prof. Harvey F. Lodish) on oncogenic signaling by cytokine receptors. In 2000 he started his laboratory at the Ludwig Institute for Cancer Research and de Duve Institute Brussels. In 2003 he became Chercheur qualifié of the FNRS. He is currently a Full Professor of Cell and Molecular biology at UCLouvain, and a Full Member of the Ludwig Institute for Cancer Research at Brussels Ludwig Cancer Research Laboratories where he directs one of the two Ludwig laboratories. He is also an internal consultant in the Service of Hematology of Cliniques Universitaires Saint-Luc.
Honors
- President, Royal Academy of Medicine of Belgium, Brussels, (Belgium) (2022-2023)
- Alexandre and Gaston Tytgat Prize for research in cancer, Belgium (2021)
- President of the Federation of European Academies of Medicine (FEAM) (2021-2024)
- Membre titulaire, Royal Academy of Medicine of Belgium, Brussels, (Belgium) (2019)
- Quinquennial Prize for 2011-2015 of the Belgian Federal Government for basic medical sciences medical research
- Welbio Investigator, Advanced Grant (2017- 2019, 2021) and Investigator, WELBIO Department, WEL Research Institute, 2022-2024)
- Honorary Member, Romanian Academy (2016)
- Pierre Stryckmans Lecture of the Belgian Society of Hematology (2014)
- Member, Academy of Medical Sciences of Romania, Bucharest (Romania) (2012)
- Prize André Matthys-Bove (Prix scientifique « Madame veuve André Matthys-Bove »), Belgium (2011)
- Maggy and Robert de Hovre Award in Immunology (Fondation Maggy et Robert de Hovre, Brussels, Belgium) (2003)
- The Medical Foundation Fellowship, Boston (U.S.A.) (1998)
- Anna Fuller Fellow in Molecular Oncology, award shared between the M.I.T. Center for Cancer Research and the Boyer Center of the Yale University (U.S.A.) (1995-1998)
- Prize of the Romanian Academy (1991)
Affiliations
- Professeur ordinaire (Full Professor), Molecular Biology and Physiology at the Université catholique de Louvain, Health Science Sector and Faculty of Medicine, Brussels, Belgium.
- Investigator, WELBIO Department, WEL Research Institute
- President of the Cell Signaling (SIGN) Pole, de Duve Institute, Université catholique de Louvain
- Professor of Cancer Signaling (Full Professorial status), Nuffield Department of Medicine, University of Oxford and Senior Group Leader, Ludwig Institute for Cancer Research Oxford.
- Member, Ludwig Institute for Cancer Research, Ludwig Cancer Research Laboratories, Brussels.
Team
Senior Investigators
Clinical Investigators
PhD Students
Research Assistants
Technical support
Administrative Support
Delineating Mpl-dependent and -independent phenotypes of Jak2 V617F-positive MPNs in vivo
Papadopoulos N, Nédélec A, Rahmani Y, Ryou H, Defour JP, Rittscher J, Royston D, Constantinescu SN
Blood (2025) 146(4):482-495
Modulation of human thrombopoietin receptor conformations uncouples JAK2 V617F-driven from cytokine-induced activation
Papadopoulos N, Pristavec A, Nédélec A, Levy G, Staerk J, Constantinescu SN
Blood (2023) blood.2022019580
Constitutive activation and oncogenicity are mediated by loss of helical structure at the cytosolic boundary of thrombopoietin receptor mutant dimers
Defour JP, Leroy E, Dass S, Balligand T, Levy G, Brett IC, Papadopoulos N, Mouton C, Genet L, Pecquet C, Staerk J, Smith SO, Constantinescu SN
Elife (2023) 12:e81521
Oncogenic CALR mutant C-terminus mediates dual binding to the thrombopoietin receptor triggering complex dimerization and activation
Papadopoulos N, Nédélec A, Derenne A, Şulea TA, Pecquet C, Chachoua I, Vertenoeil G, Tilmant T, Petrescu AJ, Mazzucchelli G, Iorga BI, Vertommen D, Constantinescu SN
Nature Comm (2023) 14(1):1881
Orientation-specific signalling by thrombopoietin receptor dimers
Staerk J, Defour JP, Pecquet C, Leroy E, Antoine-Poirel H, Brett I, Itaya M, Smith SO, Vainchenker W, Constantinescu SN
EMBO J (2011) 30(21):4398-413