Maria Veiga-da-Cunha
Institut de Duve Avenue Hippocrate 74 - B1.75.08 1200 Bruxelles
The Veiga-da-Cunha Lab focuses on understanding the impact of specific enzyme deficiencies on intermediary metabolism to find novel treatments.
Major achievements
- Recipient of an ERDERA Grant (2026)
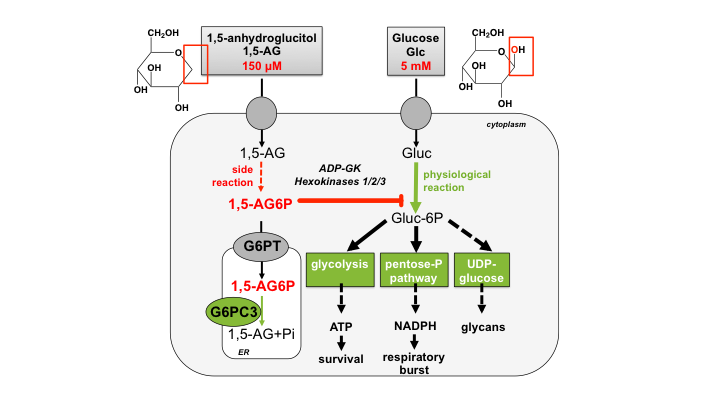
- Understanding the mechanism of neutropenia in two rare diseases (Glycogen storage disease Ib and G6PC3-deficiency).
- Repurposing of a common antidiabetic (SGLT2-inhibitors) to treat these neutropenias.
- Discovery of a new neurometabolic disease due to the deficiency of the enzyme that produces glucose-1,6-bisphosphate, a putative regulatory molecule of glucose metabolism in brain.
Recherche
Question
What are the dysfunctions in hexokinases that are at the origin of neutropenia and neurodevelopmental diseases? Can understanding them help to treat these disorders ?
Research focus
The metabolism of glucose is essential for most cells to survive and to thrive. Yet, the need for glucose is often cell-specific, and the metabolic pathways that use glucose are differently regulated due to the existence of several versions of the enzymes catalysing the critical steps of glycolysis.
Glucose phosphorylation by hexokinases (five different enzymes) is one of these critical steps. The rate of glucose utilisation has to be adjusted to the needs for downstream metabolites (ATP, pyruvate, acetyl-CoA, serine). Accordingly, hexokinases are feed-back inhibited by glucose-6-phosphate, but in a surprising way this inhibition is not yet fully understood: it is a sophisticated feature implicating the binding of glucose-6-phosphate to an allosteric site.
With her lab, Maria Veiga-da-Cunha and Emile Van Schaftingen collaborate to understand the impact of defects in the regulation of hexokinases present in inborn errors of metabolism. These can either be due to mutations in hexokinase itself or in other enzymes (including metabolite repair enzymes) or transporters that impact the production of metabolites affecting the regulation of hexokinases.
To address these questions they use two strategies : 1) an in vitro approach, where they produce and study the enzymatic properties of the recombinant enzymes and 2) a physiological one that uses cell and mouse models that mimic the enzymatic defects that are studied by metabolic analysis.
Through understanding the pathophysiological mechanism of still enigmatic inborn errors of metabolism, the Veiga-da-Cunha lab seeks to provide new treatments for the patients.
Projects
- Impact of de novo mutations in hexokinase 1 on glucose metabolism in brain
- Role of PGM2L1 in the regulation of brain glucose metabolism
- Role of the renal 1,5-anhydroglucitol transporter, SGLT5, as a major player in neutropenia due to GSD1b and G6PC3-deficiency
Projets
Maladies
Bio
Maria Veiga-da-Cunha obtained her degree in Bioengineering and Agricultural Sciences from the University of Lisbon (Portugal) and from UCLouvain (Belgium) in 1987. She obtained her D. Phil in 1990, in bacterial metabolism, in the department of biochemistry in the University of Oxford (United Kingdom). As a post-doctoral she first (1990 – 1992) joined the lab of Dr. Helena Santos, in the “Instituto de Tecnologia Quimica e Biológica (ITQB)” in Oeiras (Portugal) and learnt to use NMR techniques to study bacterial metabolism. In 1992, she moved to Brussels (Belgium) to join the lab of Prof. Emile Van Schaftingen in the de Duve Institute, where she began her training in inborn errors of metabolism and intermediary metabolism in eukaryotic cells. She became F.R.S-FNRS Research Associate and UCLouvain AssociateProfessor in 2000 and continued to closely collaborate with the Emile Van Schaftingen Lab until he become Emeritus in 2018. Since then, she has her lab but they continue to actively collaborate in most research projects.
Honors
- Robert and Maggy de Hovre prize (joint award with I. Gerin; 2000).
- Late Breaking News award at Annual Symposium of the Society for the Study of Inborn Errors of Metabolism (SSIEM) in Athens, Greece (2018).
- Alvarenga, de Piauhy 2019 prize given by the Belgium Royal Academy of Medicine - “From the bench to the bedside: a default in metabolite repair explains neutropenia in glycogen storage disease type IB (GSD1b)”.
- Late Breaking News award at Annual Symposium of the Society for the Study of Inborn Errors of Metabolism (SSIEM) in Frankfurt, Germany (2022).
Affiliations
- Group Leader, de Duve Institute
- Associate Professors, UCLouvain
- Research Associate, F.R.S. - FNRS
Equipe
Emeritus
Research Assistant
Technical supports
Pathophysiology of the Neutropenia of GSDIb and G6PC3 Deficiency: Origin, Metabolism and Elimination of 1,5-Anhydroglucitol
Veiga-da-Cunha M, Gannoun L, Dewulf J, Van Schaftingen E.
J Inherit Metab Dis (2025) 48(5):e70085.
SGLT5 is the renal transporter for 1,5-anhydroglucitol, a major player in two rare forms of neutropenia
Diederich J, Mounkoro P, Tirado HA, Chevalier N, Van Schaftingen E, Veiga-da-Cunha M
Cell Mol Life Sci (2023) 80(9):259
Impaired glucose-1,6-biphosphate production due to bi-allelic PGM2L1 mutations is associated with a neurodevelopmental disorder
Morava E, Schatz UA, Torring PM, Abbott MA, Baumann M, Brasch-Andersen C, Chevalier N, Dunkhase-Heinl U, Fleger M, Haack TB, Nelson S, Potelle S, Radenkovic S, Bommer GT, Van Schaftingen E, Veiga-da-Cunha M
Am J Hum Genet (2021) 108:1151-1160
Treating neutropenia and neutrophil dysfunction in glycogen storage disease type Ib with an SGLT2 inhibitor
Wortmann SB, Van Hove JLK, Derks TGJ, Chevalier N, Knight V, Koller A, Oussoren E, Mayr JA, van Spronsen FJ, Lagler FB, Gaughan S, Van Schaftingen E, Veiga-da-Cunha M
Blood (2020) 136:1033-1043
Failure to eliminate a phosphorylated glucose analog leads to neutropenia in patients with G6PT and G6PC3 deficiency
Veiga-da-Cunha M, Chevalier N, Stephenne X, Defour JP, Paczia N, Ferster A, Achouri Y, Dewulf JP, Linster CL, Bommer GT, Van Schaftingen E
Proc Natl Acad Sci U S A. (2019) 116:1241-1250